Wait. What? Bacterium are getting mechanical? Read more in And now for something completely different …
A must-see video. There is a reason I do not follow Dr. Faust much anymore. And as the subtitle alludes to, what next? This sends a wave of chills down my spine.
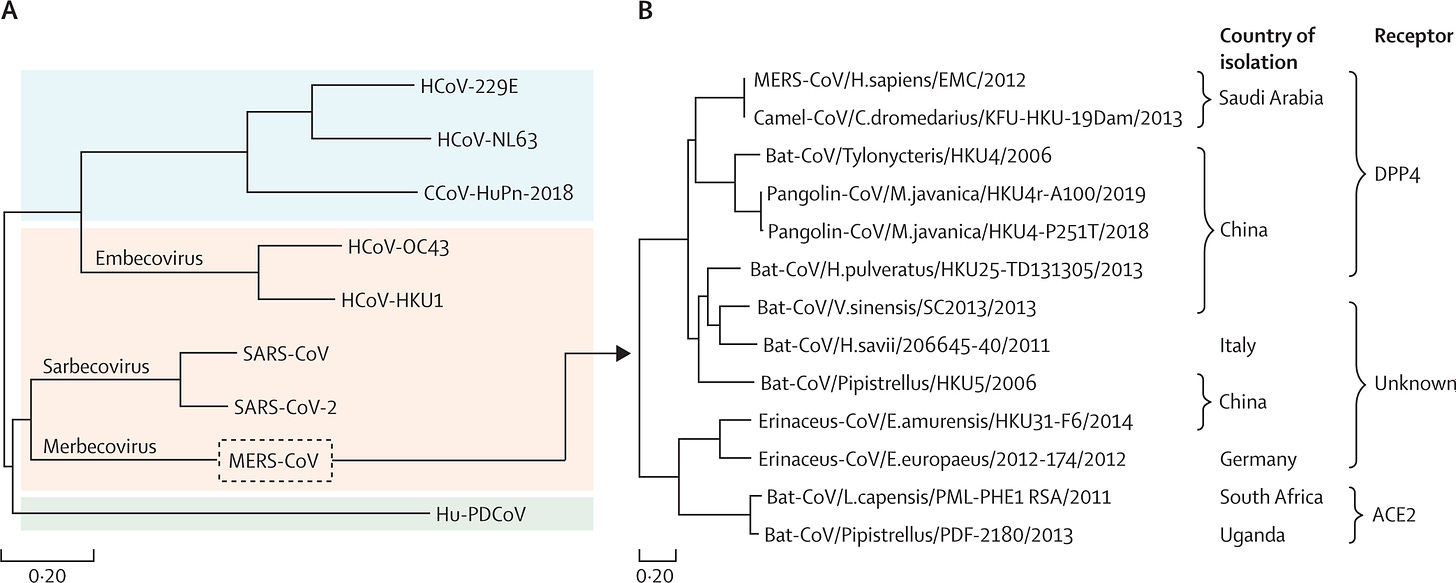
(A) Location of merbecoviruses within the coronavirus family. Only the nine coronaviruses known to cause disease in humans are shown, with genera split by colour: Alphacoronavirus (blue), Betacoronavirus (pink), and Deltacoronavirus (green). Subgenera within Betacoronavirus are also shown. Four of these nine viruses cause the common cold (HCoV-229E, HcoV-NL63, HcoV-OC43, and HcoV-HKU1), while three cause severe pneumonia (SARS-CoV, SARS-CoV-2, and MERS-CoV). Two were recently identified in a few children with pneumonia (CCoV-HuPn-2018) or fever (Hu-PDCoV) but whether these viruses will cause more widespread disease is not known. Phylogenetic analysis is based on whole-genome sequences from these disease-causing human coronaviruses. Sequences were retrieved from NCBI GenBank: SARS-CoV-2 (NC_045512.2), SARS-CoV (NC_004718.3), MERS-CoV (NC_019843.3), HCoV-OC43 (KU131570.1), HCoV-NL63 (JX504050.1), HCoV-229E (MT797634.1), HCoV-HKU1 (KF686346.1), CCoV-HuPn-2018 (MW591993.2), and Hu-PDCoV (MW685624.1). Sequences were aligned using MAFFT (version 7.511) and a maximum likelihood tree constructed using MEGA (version 11.0.13). (B) Phylogenetic analysis based on S1 domain sequences from merbecovirus spike proteins … Sequences were aligned using the MUSCLE algorithm on MEGA and a maximum likelihood tree constructed on MEGA. Country of isolation and receptor usage are indicated. In both panels, the scale bar represents the number of nucleotide (A) or amino acid (B) substitutions per site. Panel B is adapted from figure 1B in Xiong and colleagues' article.
The threat to global health security of yet another pandemic from a coronavirus remains likely because coronaviruses exhibit great genetic diversity, high rates of adaptive mutations, and readily cross species (figure A). While SARS-CoV and SARS-CoV-2 (subgenus sarbecoviruses) have been the recent focus of attention, epidemic threats of the subgenus merbecoviruses, such as MERS-CoV, deserve serious consideration. MERS-CoV was first recognised as a cause of severe zoonotic disease in 2012, with dromedary camels being the proximate source of spillover. The virus continues to circulate in camels and cause disease in humans, sometimes associated with some human-to-human transmission.
Anticipating a MERS-like coronavirus as a potential pandemic threat
But why stop there? Remember MPOX? It appears to be on the move.
Anew lineage of the mpox virus linked to efficient human-to-human transmission has been identified in the Democratic Republic of the Congo (DRC) — and the researchers behind the finding are calling for swift action to "avert another global mpox outbreak."
The Kamituga mpox outbreak in the Democratic Republic of the Congo (DRC) is caused by a novel Clade Ib lineage of Monkeypox virus (MPXV), which is distinct from previously sequenced Clade I strains in the DRC.
Sexual contact is a key mode of infection in the Kamituga outbreak, with 29% of confirmed cases being sex workers.
Genomic analysis of the MPXV strains from the Kamituga outbreak reveals a predominance of APOBEC3-type mutations, indicating recent human-to-human transmission.
The estimated time of emergence of the Kamituga MPXV lineage is around mid-September 2023, suggesting that the outbreak began recently.
Urgent measures, including reinforced surveillance, contact tracing, case management support, and targeted vaccination, are needed to contain this new Clade Ib outbreak and prevent further spread.
New mpox lineage found in DRC could help fuel another global outbreak, study warns
The actual article is here.
Shall we keep going? Maybe it’s because it is a gloomy spring day here and I am mirroring it with this newsletter but we need to examine what is up with the damn avian flu, H5N1. When a human was identified the CDC was all over the genomic sequencing. It makes me wonder why they haven’t been so forthright with the strain(s) in the cattle herd. Initially, I thought our migrating feathery friends brought it on. Not so fast. This is upsetting on so many levels. It has happened in the past too.
Experts fear that H5N1, which was only first detected in cows a few weeks ago, may have been transmitted through a type of cattle feed called “poultry litter” – a mix of poultry excreta, spilled feed, feathers, and other waste scraped from the floors of industrial chicken and turkey production plants.
In the UK and EU, feeding cows proteins from other animals has been tightly regulated since the outbreak of BSE – or ‘mad cow disease’ – 30 years ago.
Experts are unsure but fear it could be the poultry litter feed used in the US that has passed the virus to cattle.
“In the US, the feeding of poultry litter to beef cows is a known factor in the cause of botulism in cattle, and is a risk in the case of H5N1,” said Dr Steve Van Winden, Associate Professor in Population Medicine at the Royal Veterinary College.
Ground-up chicken waste fed to cattle may be behind bird flu outbreak in US cows
The CDC had another update today. Not much in it. In fact, it has been hard to any current information. And the USDA has provided little updating.
Here is a recent article on the outbreak and Emmanuel’s summary.
2) Highly Pathogenic Avian Influenza (HPAI) H5N1 strain has been detected in multiple dairy cattle herds across several US states, which is unusual as avian influenza rarely infects cattle. Genetic analysis shows the virus belongs to the Eurasian H5N1 goose/Guangdong lineage.
3) It is suspected to have been introduced by wild migratory birds.
Infected cows showed symptoms like decreased milk production.
Genomic analysis found some genetic variation in the HA and NA genes between avian, mammal and human viruses, but genetic distance is small4) ...indicating strains are similar.
In HA, birds showed positive selective pressure while mammals and humans did not. In NA, mammals showed positive pressure while birds and humans did not.
This suggests birds remain the primary host but virus is accumulating genetic5) ...variability in mammals through drift, enhancing hazard potential over time.
Swift surveillance, response and biosecurity are needed to contain the outbreak and prevent further spread between animals and potential spillover to humans.
Oops! I forgot about that COVID-19 thing. We had an update last Friday.
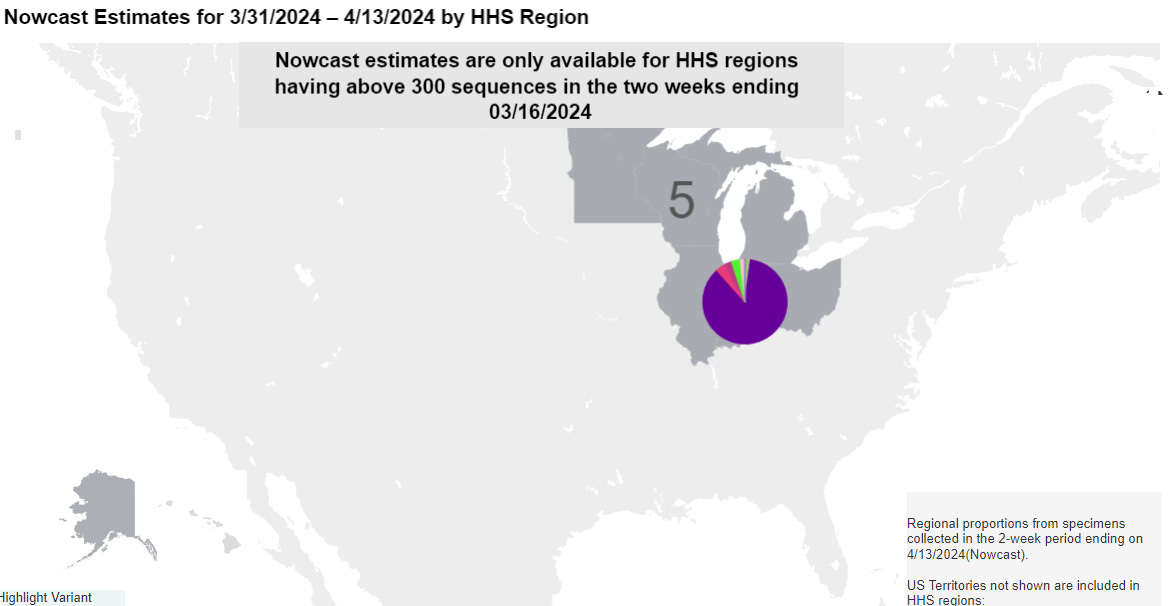
Well, we are up 4 flavors of JN.1.x. And here is the dismal part of the analysis. Yep. One region …
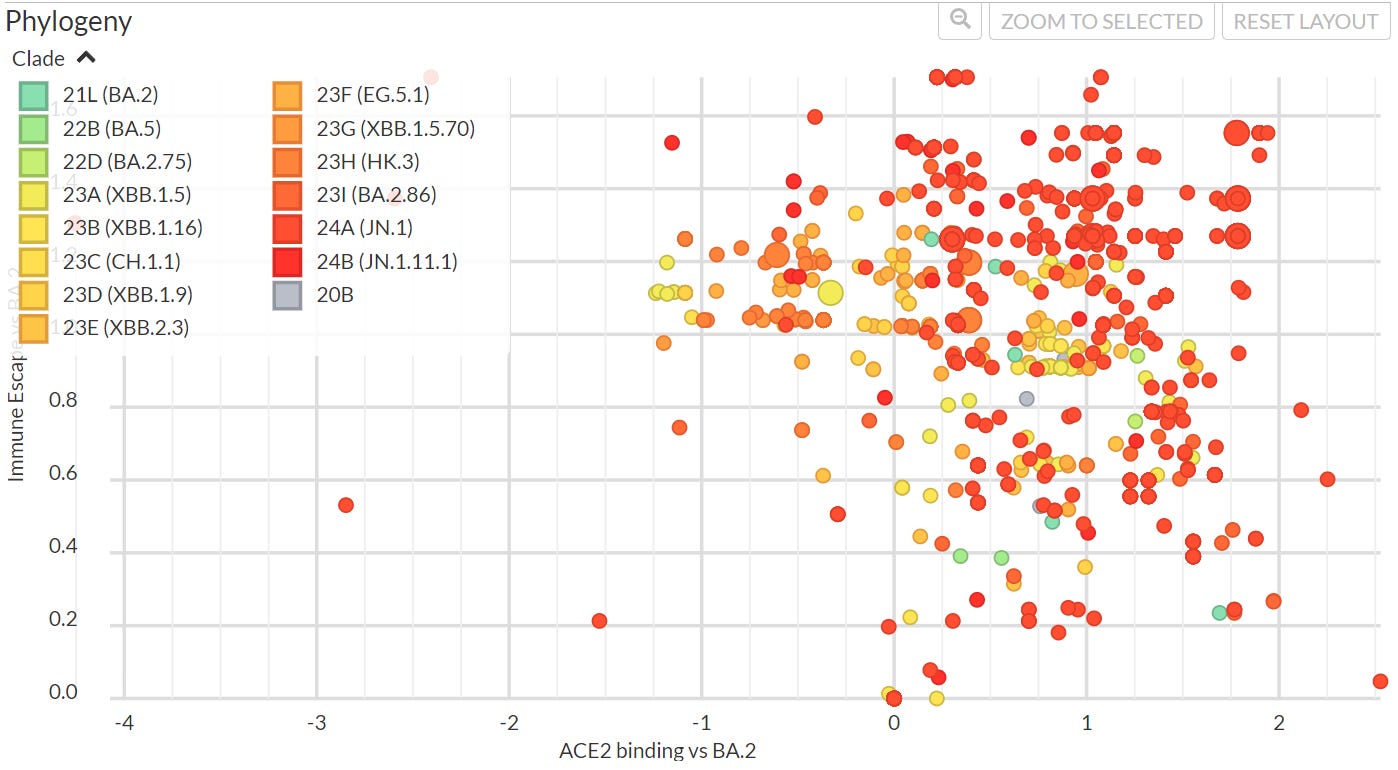
What that does not show is that other variants have caused a stir. TACT has been writing about KP.3 lately, and it has an incredible GA, ~170%. Here are the latest from NEXTSTRAIN.org. Notice there is quite a creep towards the transmission component (ACE2 binding) of the virus.
Oh shoot, almost forgot measles … There should be another CDC update tomorrow. Here is the most recent.
Measles is a highly infectious febrile rash illness and was declared eliminated in the United States in 2000. However, measles importations continue to occur, and U.S. measles elimination status was threatened in 2019 as the result of two prolonged outbreaks among undervaccinated communities in New York and New York City. To assess U.S. measles elimination status after the 2019 outbreaks and to provide context to understand more recent increases in measles cases, CDC analyzed epidemiologic and laboratory surveillance data and the performance of the U.S. measles surveillance system after these outbreaks. During January 1, 2020–March 28, 2024, CDC was notified of 338 confirmed measles cases; 97 (29%) of these cases occurred during the first quarter of 2024, representing a more than seventeenfold increase over the mean number of cases reported during the first quarter of 2020–2023. Among the 338 reported cases, the median patient age was 3 years (range = 0–64 years); 309 (91%) patients were unvaccinated or had unknown vaccination status, and 336 case investigations included information on ≥80% of critical surveillance indicators. During 2020–2023, the longest transmission chain lasted 63 days. As of the end of 2023, because of the absence of sustained measles virus transmission for 12 consecutive months in the presence of a well-performing surveillance system, U.S. measles elimination status was maintained. Risk for widespread U.S. measles transmission remains low because of high population immunity. However, because of the increase in cases during the first quarter of 2024, additional activities are needed to increase U.S. routine measles, mumps, and rubella vaccination coverage, especially among close-knit and undervaccinated communities. These activities include encouraging vaccination before international travel and rapidly investigating suspected measles cases.
Here is a recent article that is a short read on measles. Sobering.
“What was surprising about 2024 is that we’ve seen a significant increase,” John Brownstein, an epidemiologist at Boston Children’s Hospital, tells ABC News’ Mary Kekatos. “It’s an alarming number, because it indicates a trend going in the wrong direction for us, a virus that we have successfully controlled, a virus that we successfully have an effective vaccine for.”
U.S. Measles Cases Are Rising in 2024, CDC Warns
Okay, I cry uncle.
The Covid Wire
Sometimes folks think I go over the top in avoiding situations that could lead to a COVID-19 infection. Well, here is more data indicating that avoidance at all costs is worth it.
We observed that SARS-CoV-2 spike protein interrupts p53-MDM2 protein interaction but did not detect SARS-CoV-2 spike bound with p53 protein in the cancer cells. We further observed that SARS-CoV-2 spike suppresses p53 transcriptional activity in cancer cells including after nutlin exposure of wild-type p53-, spike S2-expressing tumor cells and inhibits chemotherapy-induced p53 gene activation of p21(WAF1), TRAIL Death Receptor DR5 and MDM2. The suppressive effect of SARS-CoV-2 spike on p53-dependent gene activation provides a potential molecular mechanism by which SARS-CoV-2 infection may impact tumorigenesis, tumor progression and chemotherapy sensitivity. In fact, cisplatin-treated tumor cells expressing spike S2 were found to have increased cell viability as compared to control cells.
Let’s stay on the subject of immunodeficiency and COVID-19. The common call for new, aggressive treatment is highlighted in this work.
Because immunocompromised individuals generate poor vaccine or infection-derived immune responses, this may also predispose them for becoming transient reservoirs for SARS-CoV-2 variant evolution.
In patients with severe hematologic-oncology/transplant (S-HT), severe autoimmune/B cell deficiency (S-A), non-severe (NS), and non-immunocompromised controls, the authors found that upper airway viral RNA decay was predicted by immunocompromisation status, with the S-HT group showing a significant delay in viral clearance.
Although these observations should be confirmed in other patient demographics and in larger studies, these findings suggest that more aggressive interventions may be needed to treat patients with S-HT and S-A. These aggressive treatment strategies could include combination therapy of immunologic strategies (e.g., passively infused monoclonal antibodies) and orally administered direct-acting antivirals (e.g., Mpro inhibitor Paxlovid) to evaluate whether combination strategies lead to more rapid clearance of SARS-CoV-2 and reduce the risk of mutational escape from single-agent therapies. These findings also highlight that additional therapies, such as next-generation orally administered direct-acting antivirals, are needed to suppress SARS-CoV-2 replication and dampen viral evolution in severely immunocompromised patients. Optimizing viral-targeting combination therapies may also result in more effective treatments in this vulnerable and immunodeficient population.
Mutations spike in a reservoir of compromised immunity
On the Long Covid front, this came out since my last newsletter. A slight breath of optimism in the article.
Scientists have started to unlock the mysteries of Long Covid – and have identified five different forms of the illness.
British researchers have shed light on why, in some people who caught the Covid-19 virus, the immune system continues to attack itself and cause debilitating fatigue and brain fog. The team led by Imperial College London tracked 660 hospitalised Covid patients and detected telltale proteins in blood samples that indicate what is happening to the body.
The most in depth ever study of the little-understood syndrome indicates some existing drugs for conditions such as arthritis could work against Long Covid - and will now become the focus of new human trials.
Long Covid discovery reveals it's actually five different illnesses - and could lead to a cure
Read the research article here.
MAPS
I have some good news concerning MAPS. The memory leak has been plugged and the code has been cleaned up. I should have a new forecast out this weekend. I am currently trying to add county-level wastewater data to the MAPS initialization. Quite tedious but the forecast accuracy should improve dramatically.
The Covid Xfeed
I will drop a bunch of these in the next newsletter, you’ve probably had enough for the day.
And now for something completely different …
Come on, you have been thinking about this all your life …
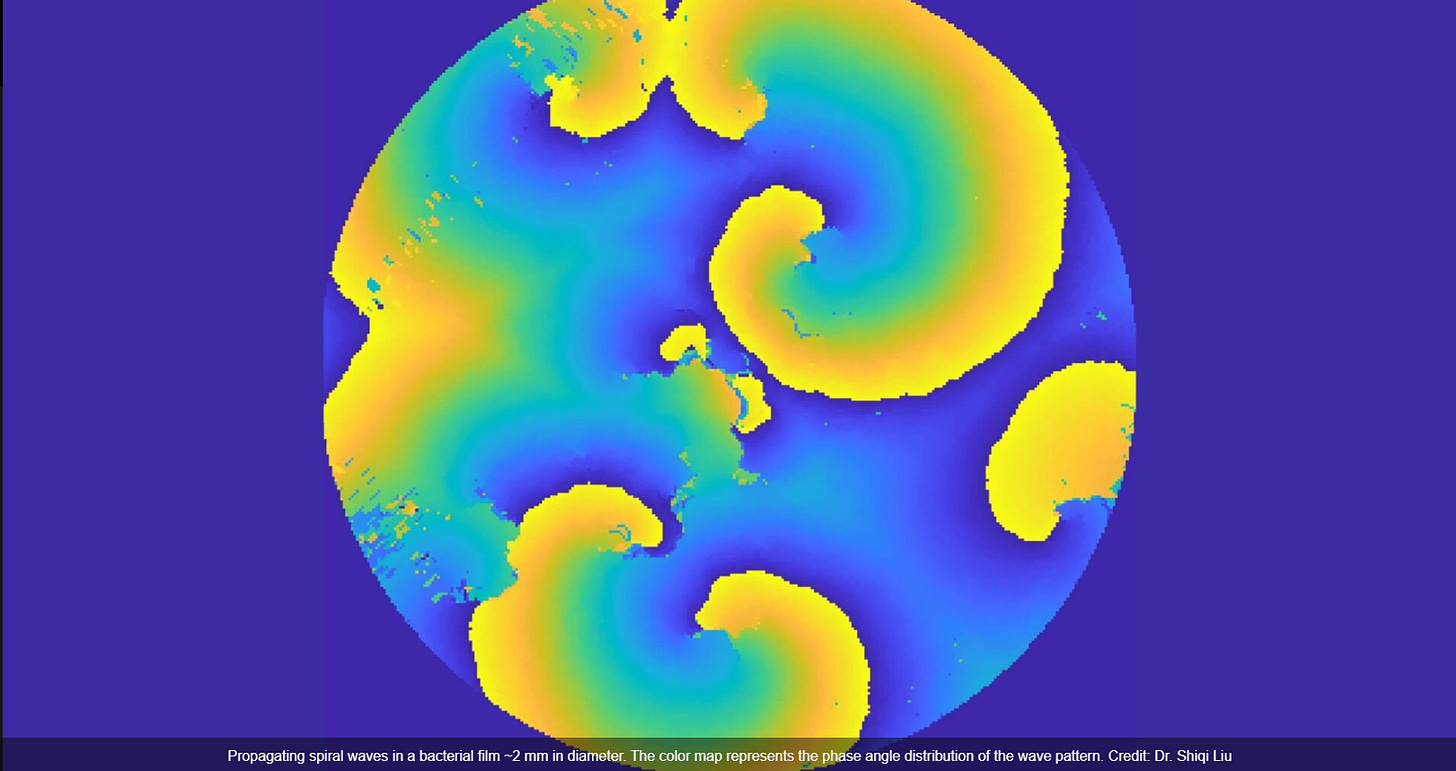
A new study by researchers from The Chinese University of Hong Kong has reported the emergence of mechanical spiral waves in bacterial matter.
Spiral waves are commonly seen in artificial and natural systems (such as the heart). These emerge from interactions of neighboring elements, such as cardiac cells in the case of the heart. These spiral waves can have varying effects, sometimes leading to life-threatening conditions like fibrillation in the heart.
The researchers believe that the pilus motors not only serve as mechanical actuators but also as sensors. This means that they can detect mechanical stimuli in the environment in the environment, which allows for synchronized movements within bacterial populations.
"We believe that the coordination or coupling of pilus activities allows bacterial populations to control large-scale tension forces and may influence their dispersal," explained Dr. Wu.
Therefore, understanding spiral waves can help to understand the behavior of bacterial species.
Additionally, stationary spiral waves are found in many diverse systems. "The wave pattern in the pilus-powered bacterial matter may, therefore, provide a tractable mechanical analog for investigating the origin and control of stable spiral waves in diverse living systems, such as cardiac tissues," explained Dr. Liu.
Spiraling insights: Scientists observe mechanical waves in bacterial communities
Here is the actual research article.
Theoretical modelling reveals that the spiral tension waves result from non-reciprocity in cell–cell interactions. Our findings reveal a mechanism of large-scale force regulation in bacterial world and may shed light on the emergent mechanics of biofilms and microbiomes. Pilus-driven bacterial living matter also provides a mechanical active medium for studying electrical or chemical spiral waves in living systems.
A big thing that went largely unnoticed.
And another big thing.
Stay safe and help those who need it most.
take care,
Joe
*A special note: this is a free newsletter. I spend 4 hours a day just reading and another couple of hours putting together this newsletter. Donations out of kindness are deeply appreciated. All donations go toward the purchase of another server that will allow my modeling work to go global with a small percentage toward climate causes. You can go to my substack homepage if you wish to donate.
Where do viruses hide in the human body? | The BMJ
Understanding immunity and viruses through the John Snow Project
Variant tracking at the CDC
Infections at WHN (updated!)
The neuroinvasiveness, neurotropism, and neurovirulence of SARS-CoV-2: Trends in Neurosciences (cell.com)







Thank you! I do not know what was up yesterday but I could not get some usda pages to load.
This might be helpful for some? https://www.aphis.usda.gov/sites/default/files/notice-24-06.pdf