It’s too hot damn hot in here Queen Bee. I am going to go out and buzz some damn humans. Read more in And now for something completely different …
I will keep this a bit brief today. I got the model running the next 4-month forecast and am working on some post-processing issues that have arisen on the new server. I will try to wrap up the forecast tomorrow.
Let’s start with the shiny virus of the day, H5N1. Beware, many other bright objects have been distracting me lately. I will briefly mention these later. So let us proceed the the Xfeed.
The Covid Xfeed
I cannot do justice to summarizing some important contributions to our knowledge of H5N1. I will defer to the brilliant Emmanuel for this. He posted an elegant multi-part summary in the last few days on various aspects of it. He kept indicating “LAST UPDATE!” but was to impassioned to stop. Thanks for writing my newsletter.
Emmanuel
𝗛5𝗡1 𝗟𝗔𝗦𝗧 𝗨𝗣𝗗𝗔𝗧𝗘 !
(🧵 𝘗𝘈𝘙𝘛 1)
"𝘗𝘳𝘦𝘭𝘪𝘮𝘪𝘯𝘢𝘳𝘺 𝘳𝘦𝘱𝘰𝘳𝘵 𝘰𝘯 𝘨𝘦𝘯𝘰𝘮𝘪𝘤 𝘦𝘱𝘪𝘥𝘦𝘮𝘪𝘰𝘭𝘰𝘨𝘺 𝘰𝘧 𝘵𝘩𝘦 2024 𝘏5𝘕1 𝘪𝘯𝘧𝘭𝘶𝘦𝘯𝘻𝘢 𝘈 𝘷𝘪𝘳𝘶𝘴 𝘰𝘶𝘵𝘣𝘳𝘦𝘢𝘬 𝘪𝘯 𝘜.𝘚. 𝘤𝘢𝘵𝘵𝘭𝘦 (𝘱𝘢𝘳𝘵 1)"
2) H/t @MichaelWorobey @PeacockFlu and colleagues
BACKGROUND :
Reassortment of the virus with local avian strains has generated diverse genotypes. virological.org/t/preliminary-…
Preliminary report on genomic epidemiology of the 2024 H5N1 influenza A virus outbreak in U.S. cattle (Part 1 of 2)Due to length constraints on virological.org posts, we are splitting this report into two parts that should be read as a single report. This is Part 1, containing Background, Data, Methods, and Findi…https://virological.org/t/preliminary-report-on-genomic-epidemiology-of-the-2024-h5n1-influenza-a-virus-outbreak-in-u-s-cattle-part-1-of-2/970
3) The virus has a high propensity to infect mammals, including domestic cats, foxes, mink, and pinnipeds. Mammalian infections lead to severe disease and quick adaptation. Bird-to-mammal transmission is uncertain.
4) An outbreak of H5N1 was detected in dairy cattle in the USA, causing reduced milk production, severe mastitis, and mild respiratory disease. The full severity is unknown.
5) FINDINGS
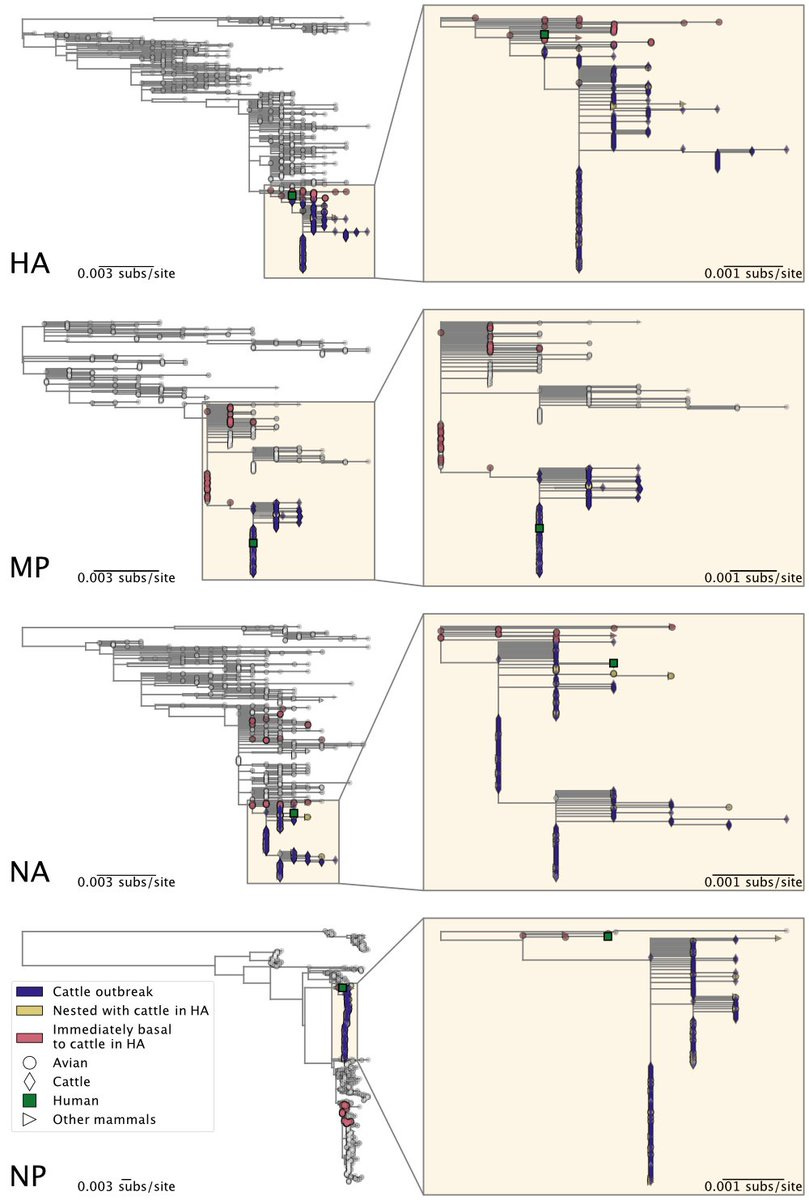
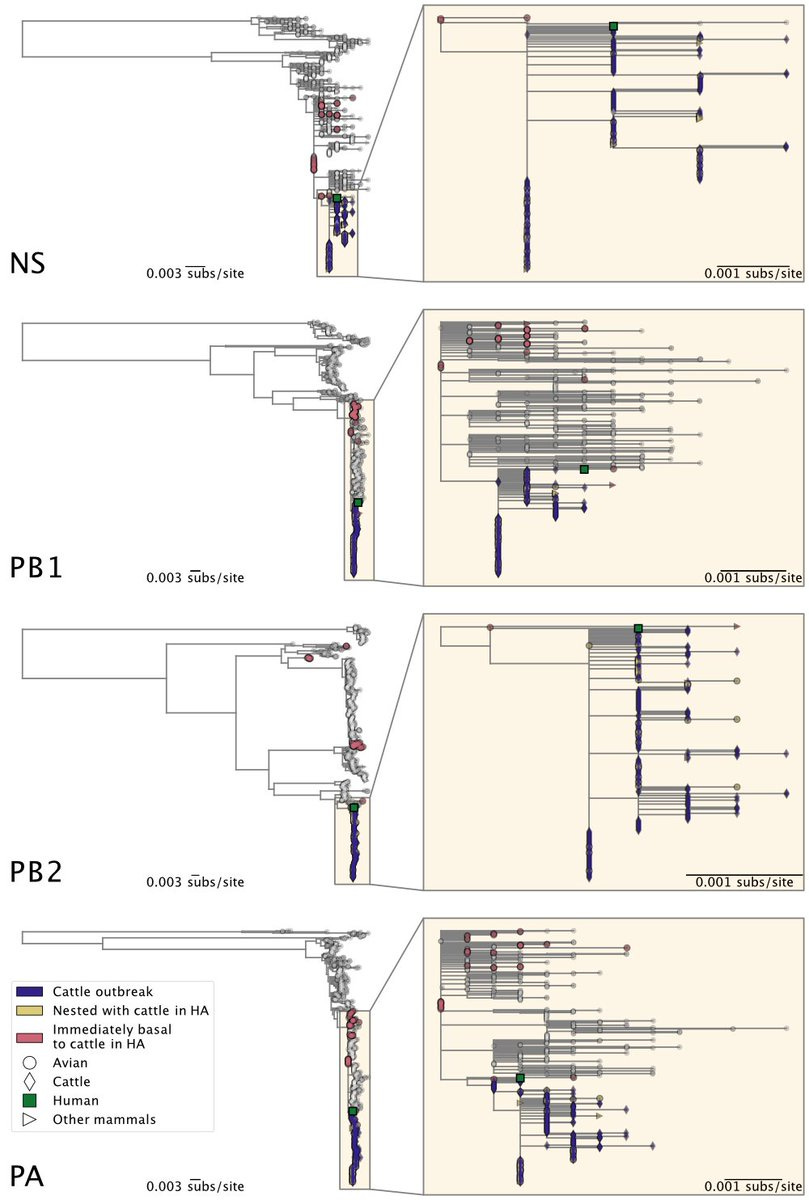
A reassortment event occurred within the North American avian H5N1 2.3.4.4b viruses before the start of the cattle outbreak. The cattle sequences are all Genotype B3.13, which is a reassortant between the Eurasian panzootic H5N1 genotype and low pathogenicity ...
6) ... North American genotypes.
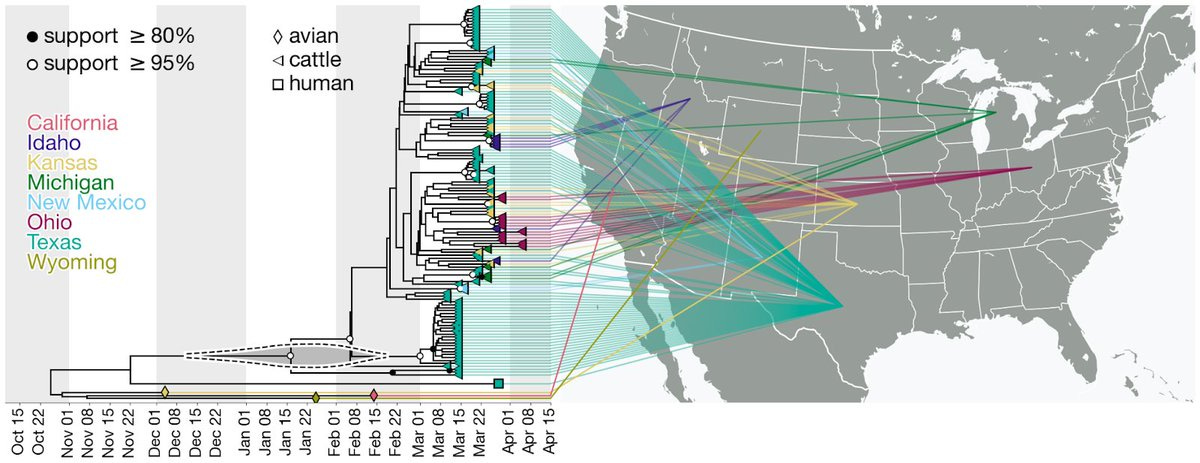
The cattle outbreak likely originated from a single introduction of H5N1 into cows and spread among cattle. The viruses sampled from cattle form a monophyletic clade in each genome segment, indicating cattle-to-cattle transmission.
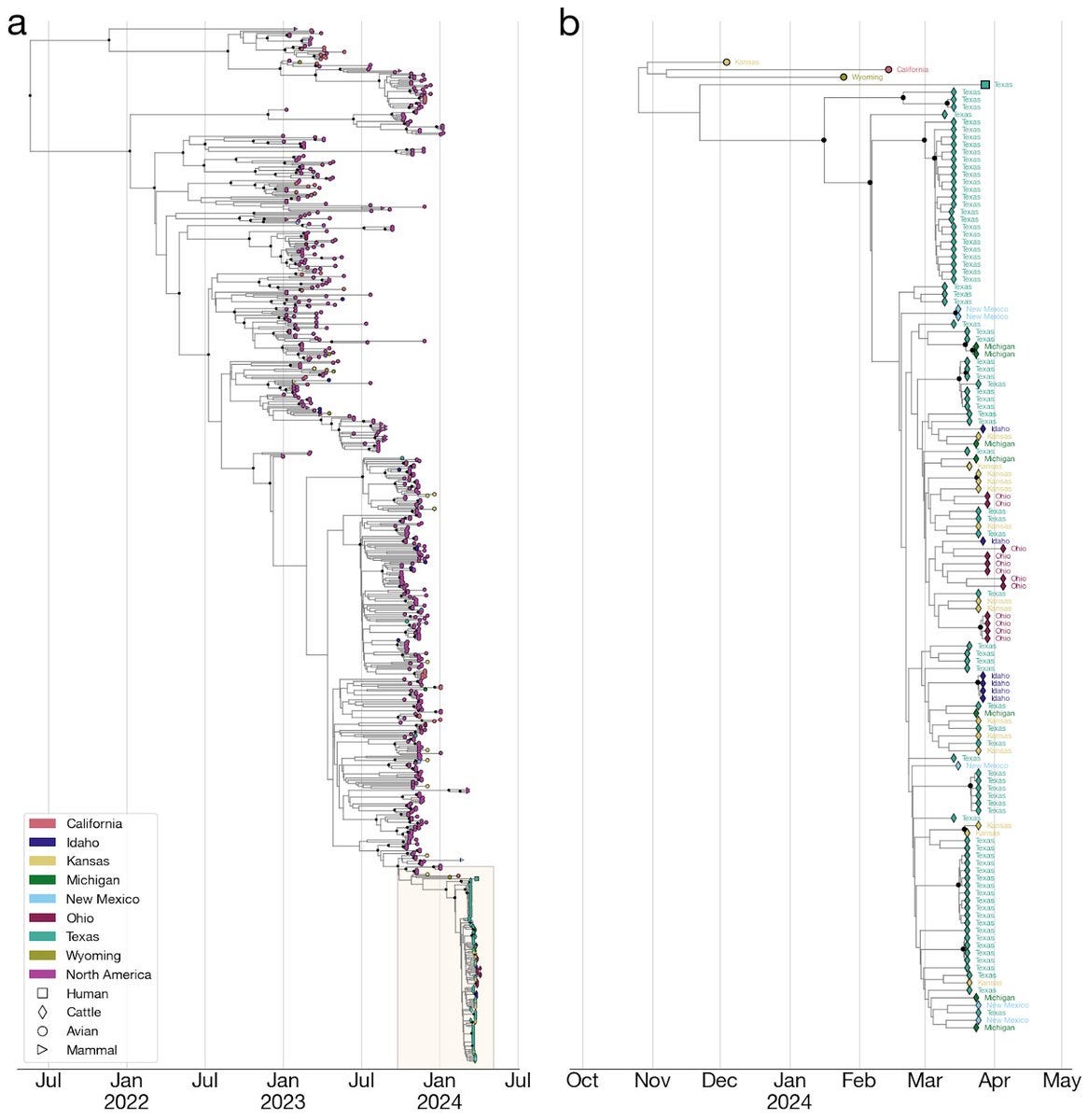
7) The H5N1 outbreak in cattle may have gone undetected and unidentified for an extended period of time. Molecular clock estimates suggest that the virus may have been circulating in cattle for up to 5 months before it was identified.
8) The cattle outbreak may have originated in Texas, where the first ill and infected cattle were reported. The phylogenetic tree shows basal diversity sampled in Texas, but further analysis is needed to determine the movement of the virus across states.
9) The cattle H5N1 clade has several putatively adaptive substitutions in the polymerase complex, which are necessary for the virus to infect mammals. These substitutions indicate adaptation to use mammalian versions of a host protein called ANP32.
10) There is minimal evidence for a different selective regime acting within the cattle H5N1 virus clade compared to avian H5N1 viruses, except for a modest increase in the intensity of selection in the PA segment.
𝗛5𝗡1 𝗟𝗔𝗦𝗧 𝗨𝗣𝗗𝗔𝗧𝗘 !
(🧵 𝘗𝘈𝘙𝘛 2)
"𝘗𝘳𝘦𝘭𝘪𝘮𝘪𝘯𝘢𝘳𝘺 𝘳𝘦𝘱𝘰𝘳𝘵 𝘰𝘯 𝘨𝘦𝘯𝘰𝘮𝘪𝘤 𝘦𝘱𝘪𝘥𝘦𝘮𝘪𝘰𝘭𝘰𝘨𝘺 𝘰𝘧 𝘵𝘩𝘦 2024 𝘏5𝘕1 𝘪𝘯𝘧𝘭𝘶𝘦𝘯𝘻𝘢 𝘈 𝘷𝘪𝘳𝘶𝘴 𝘰𝘶𝘵𝘣𝘳𝘦𝘢𝘬 𝘪𝘯 𝘜.𝘚. 𝘤𝘢𝘵𝘵𝘭𝘦 (𝘱𝘢𝘳𝘵 2)"
virological.org/t/preliminary-…
Preliminary report on genomic epidemiology of the 2024 H5N1 influenza A virus outbreak in U.S. cattle (Part 2 of 2)Due to length constraints on virological.org posts, we are splitting this report into two parts that should be read as a single report. This is Part 2, containing Findings sections 6 through 9, Concl…https://virological.org/t/preliminary-report-on-genomic-epidemiology-of-the-2024-h5n1-influenza-a-virus-outbreak-in-u-s-cattle-part-2-of-2/971
2) The original cattle H5N1 virus's hemagglutinin (HA) protein was not adapted to bind to human-like receptors. The closest-related sequences to the cattle H5N1 HA came from wild birds, indicating no pre-adaptation to mammalian receptors.
3) Mutations in HA and the polymerase have been observed in small clades from cattle and cats, but their significance is unclear. Without changes in HA affecting receptor binding, the risk of human transmission is low.
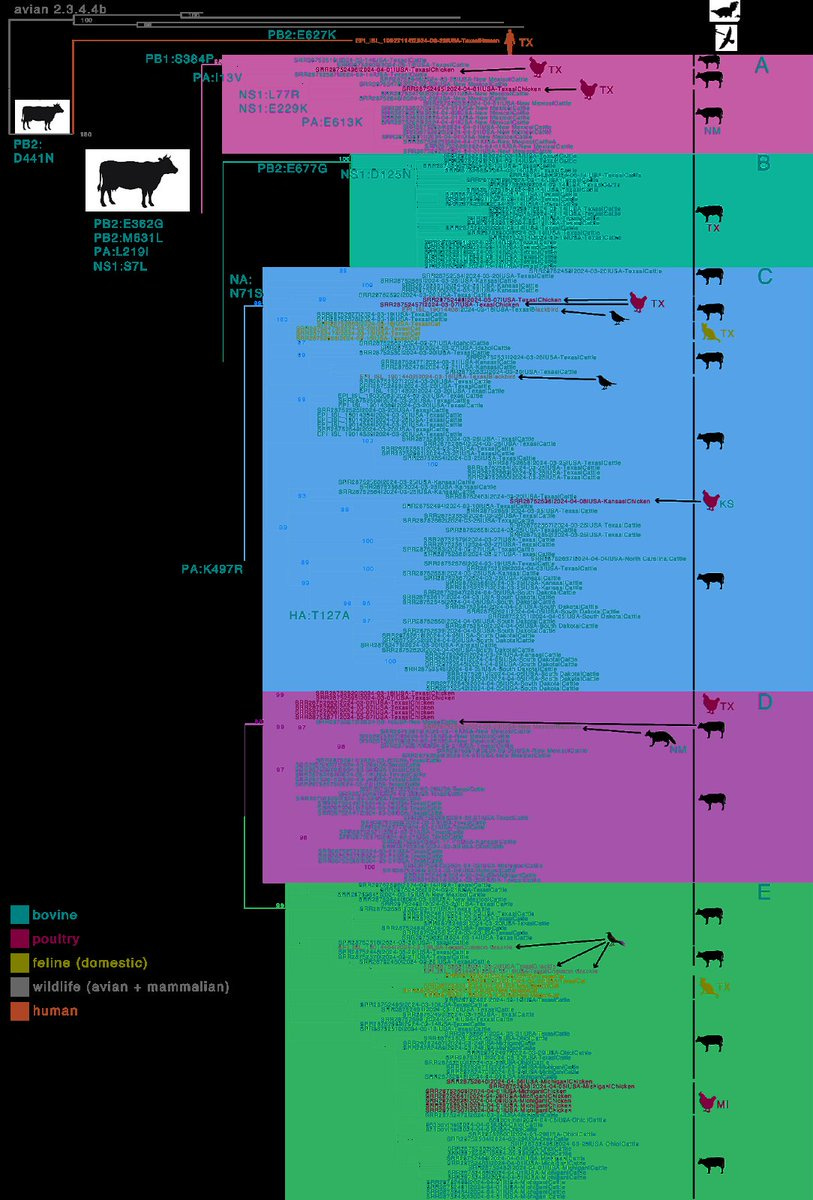
H5N1 has transmitted from cattle to wild birds, poultry ...
4) ...cats, and other species. Sequences from various hosts are nested within the cattle clade, suggesting spillback from cows. Spillbacks to domestic cats have occurred, likely due to raw milk consumption.
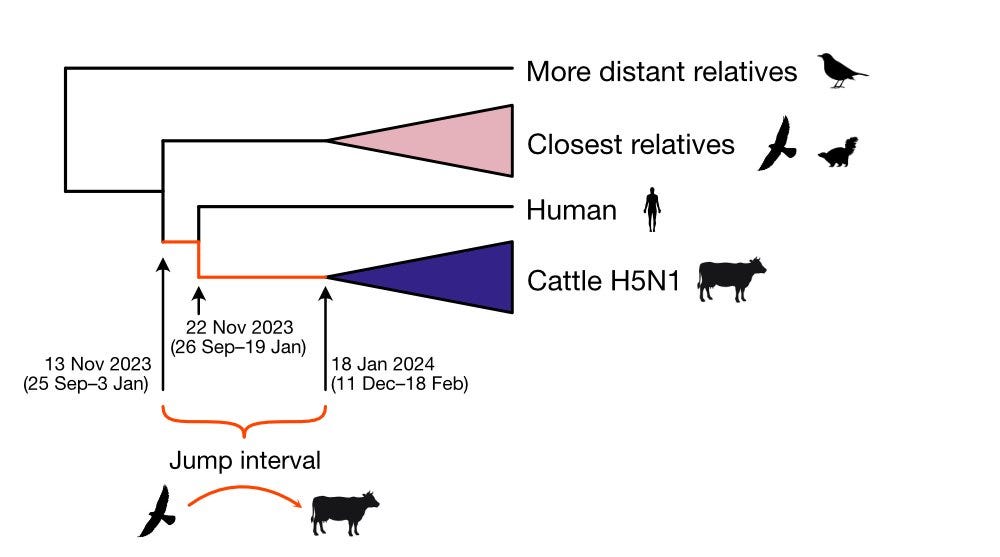
5) The tMRCA of the cattle clade and the human virus is estimated to be November 2023. The human sequence has unique amino acid changes not found in cattle or basal avian/wildlife sequences.
The transmission chain is unclear, but a direct avian spillover is unlikely.
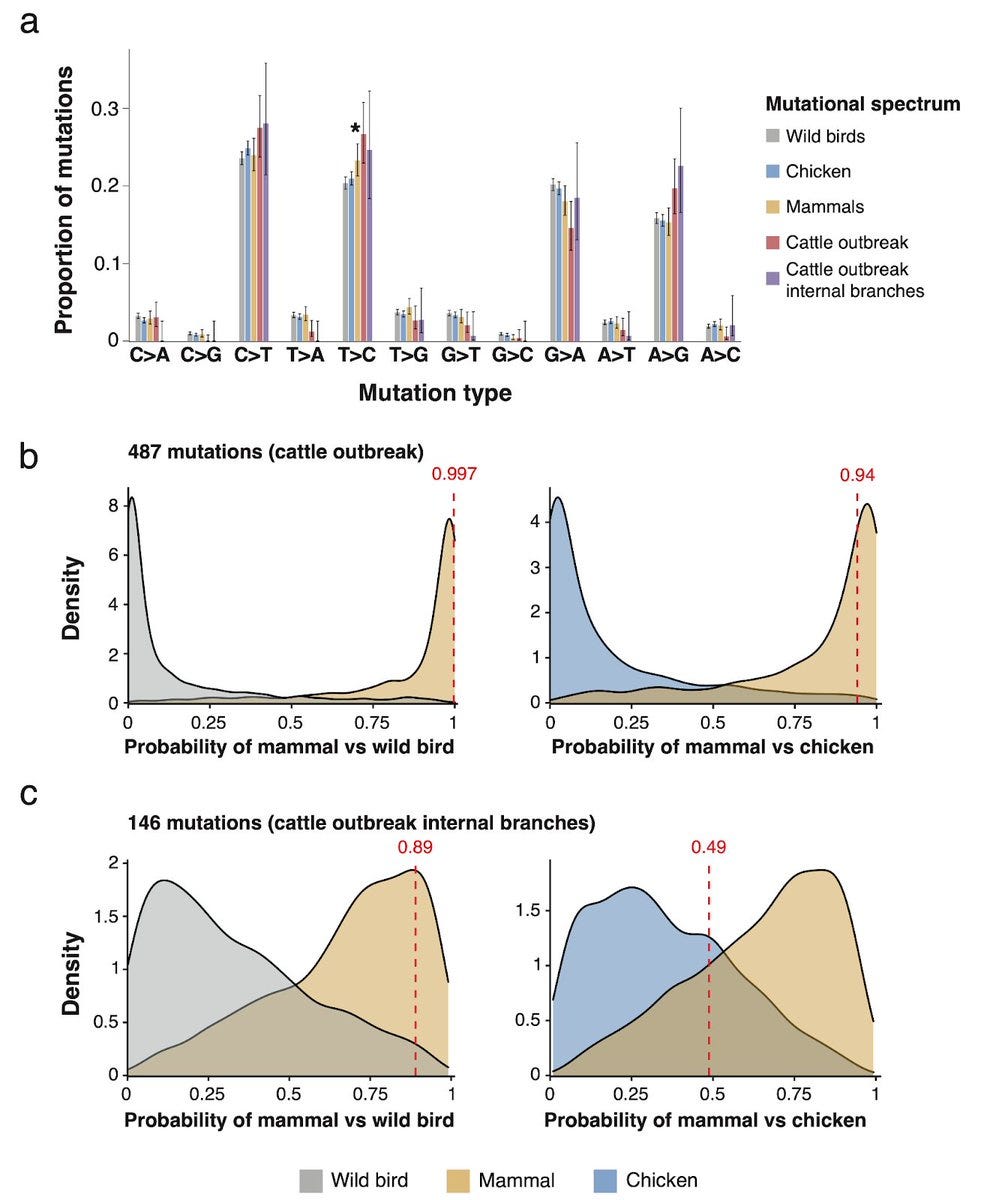
6) Mutational signatures support sustained transmission within cattle, with elevated T-to-C mutations in mammals. The mutational spectrum of the cattle outbreak indicates transmission among mammals.
The mutations are more likely to have been generated under the mammal spectrum.
7) Overall, cattle H5N1 has the potential to transmit to other species, including humans, but sustained human transmission risk is currently low.
𝗛5𝗡1 𝗟𝗔𝗦𝗧 𝗨𝗣𝗗𝗔𝗧𝗘 !
(🧵 𝘗𝘈𝘙𝘛 3 : 𝘛𝘩𝘦 𝘙𝘐𝘚𝘒𝘚)
"H5N1 SCENARIO-based Human Health Risk Assessment for the United States as of [5/1/2024]"
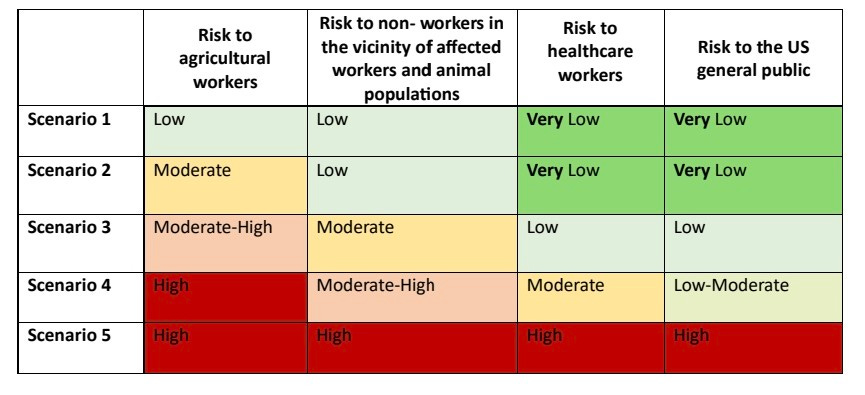
2) The risk assessment report considers 5 possible SCENARIOS for how the H5N1 outbreak in cattle could evolve and the corresponding risks to human health.
Currently the outbreak is judged to be between SCENARIOS 2 and 3, with widespread cases in cattle and some spread to ...
3) ...other animals like cats, but no human-to-human transmission reported so far.
SCENARIO 1 assumes minimal spread in animals and low risk to all human populations.
SCENARIO 2 involves widespread cattle spread and some cow-to-human transmission ...
4) ...posing moderate risk to farmers and low risk to others.
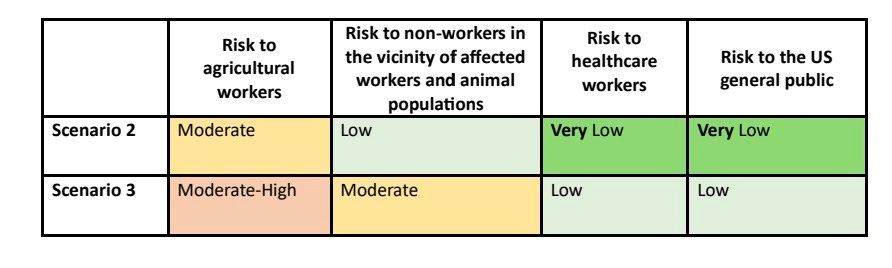
SCENARIO 3 adds potential for animal mixing and human transmission among close contacts of infected people or farmers. Risk increases moderately.
5) SCENARIO 4 sees more human infections via animals and limited human-to-human transmission, increasing risk levels further especially for farmers, their contacts and healthcare workers.
SCENARIO 5 is full human-to-human transmission ...
6) ... posing high risk to all populations as the virus would be very difficult to contain.
Risk is assessed based on transmission factors, health impacts, testing levels, and response measures like PPE use, tracing, quarantine and vaccine development.
7) Ongoing recommendations focus on animal surveillance and separation, protective equipment for workers, information sharing, and public health preparations for potential escalation.
Source :
H/t @dr_kkjetelina @cmyeaton
This part is extremely important! I will send the next day’s posts shortly. By the time you are done reading, you will know more than 95% of the human race about H5N1.
𝗛5𝗡1 𝗟𝗔𝗦𝗧 𝗨𝗣𝗗𝗔𝗧𝗘 !
(🧵 𝘗𝘈𝘙𝘛 4)
"𝘛𝘩𝘦 𝘢𝘷𝘪𝘢𝘯 𝘢𝘯𝘥 𝘩𝘶𝘮𝘢𝘯 𝘪𝘯𝘧𝘭𝘶𝘦𝘯𝘻𝘢 𝘈 𝘷𝘪𝘳𝘶𝘴 𝘳𝘦𝘤𝘦𝘱𝘵𝘰𝘳𝘴 𝘴𝘪𝘢𝘭𝘪𝘤 𝘢𝘤𝘪𝘥 (𝘚𝘈)-α2,3 𝘢𝘯𝘥 𝘚𝘈-α2,6 𝘢𝘳𝘦 𝘸𝘪𝘥𝘦𝘭𝘺 𝘦𝘹𝘱𝘳𝘦𝘴𝘴𝘦𝘥 𝘪𝘯 𝘵𝘩𝘦 𝘣𝘰𝘷𝘪𝘯𝘦 𝘮𝘢𝘮𝘮𝘢𝘳𝘺 𝘨𝘭𝘢𝘯𝘥"
2) Reference
Avian influenza A virus (IAV) infections were recently detected in dairy cows in the US. Cattle are not typically considered susceptible hosts for IAV. biorxiv.org/content/10.110…
The avian and human influenza A virus receptors sialic acid (SA)-α2,3 and SA-α2,6 are widely expressed in the bovine mammary glandAn outbreak of H5N1 highly pathogenic influenza A virus (HPIAV) has been detected in dairy cows in the United States. Influenza A virus (IAV) is a negative-sense, single-stranded, RNA virus that has n…https://www.biorxiv.org/content/10.1101/2024.05.03.592326v1
3) IAV receptors are sialic acids linked to galactose in either an α2,3 or α2,6 linkage. Human IAVs prefer the α2,6 receptor, avian IAVs the α2,3 receptor.
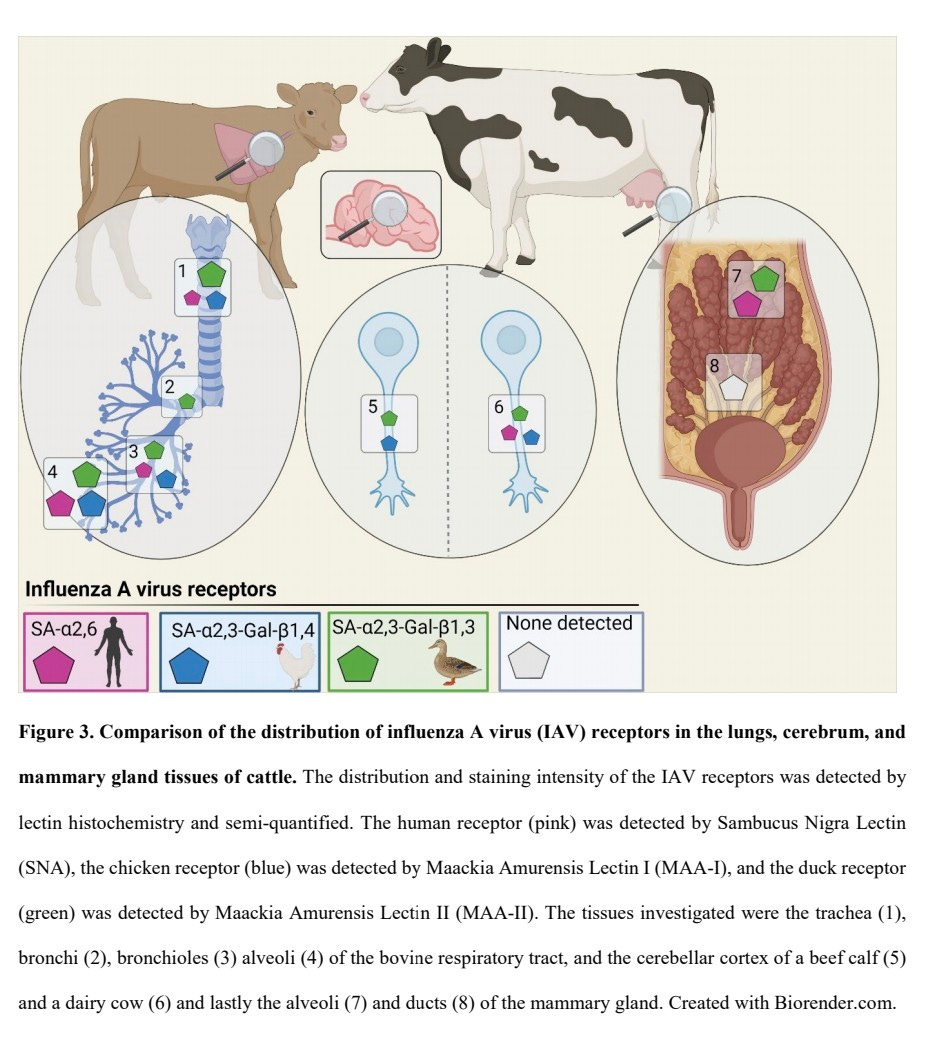
The study investigated expression of IAV receptors (α2,3, α2,6, α2,3-β1,4, α2,3-β1,3) in bovine respiratory tract ...
4) ...cerebrum, and mammary glands using lectin histochemistry.
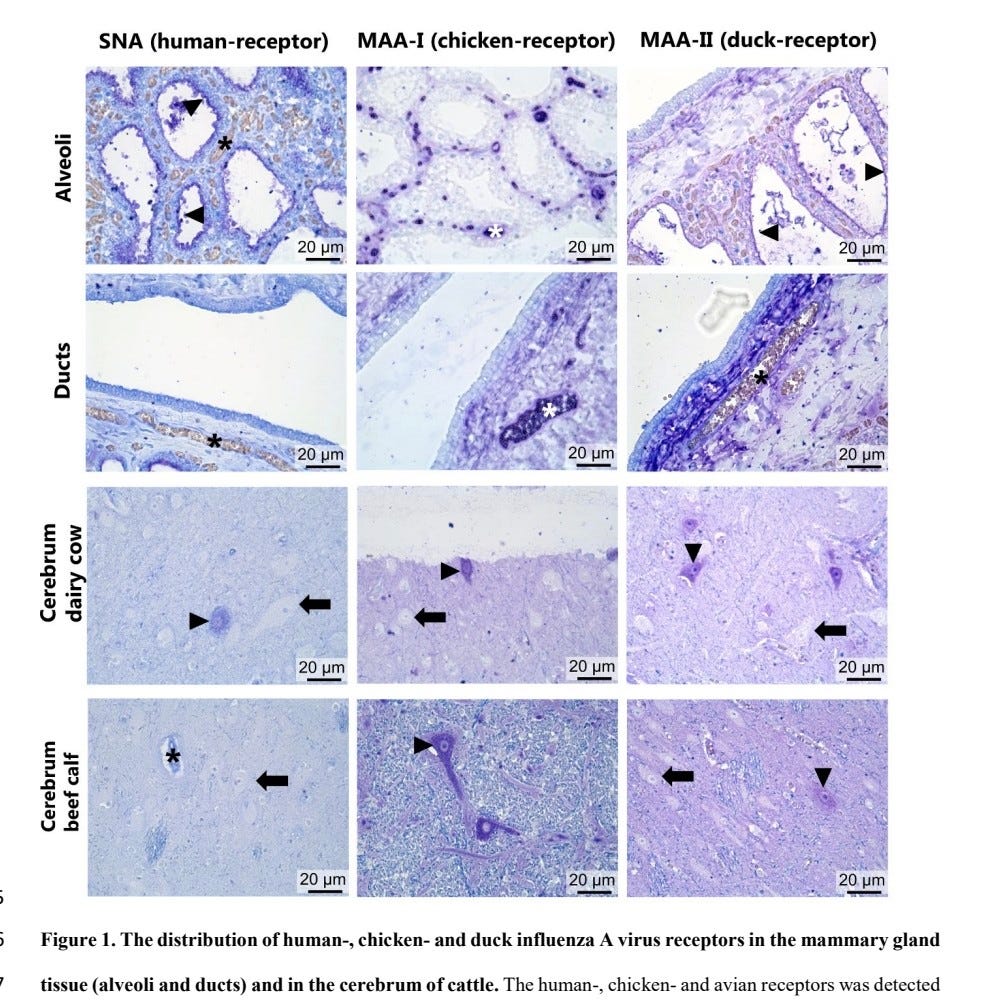
The duck (α2,3-β1,3) and human (α2,6) receptors were widely expressed in mammary gland alveoli, but not ducts. The chicken (α2,3-β1,4) receptor was not detected.
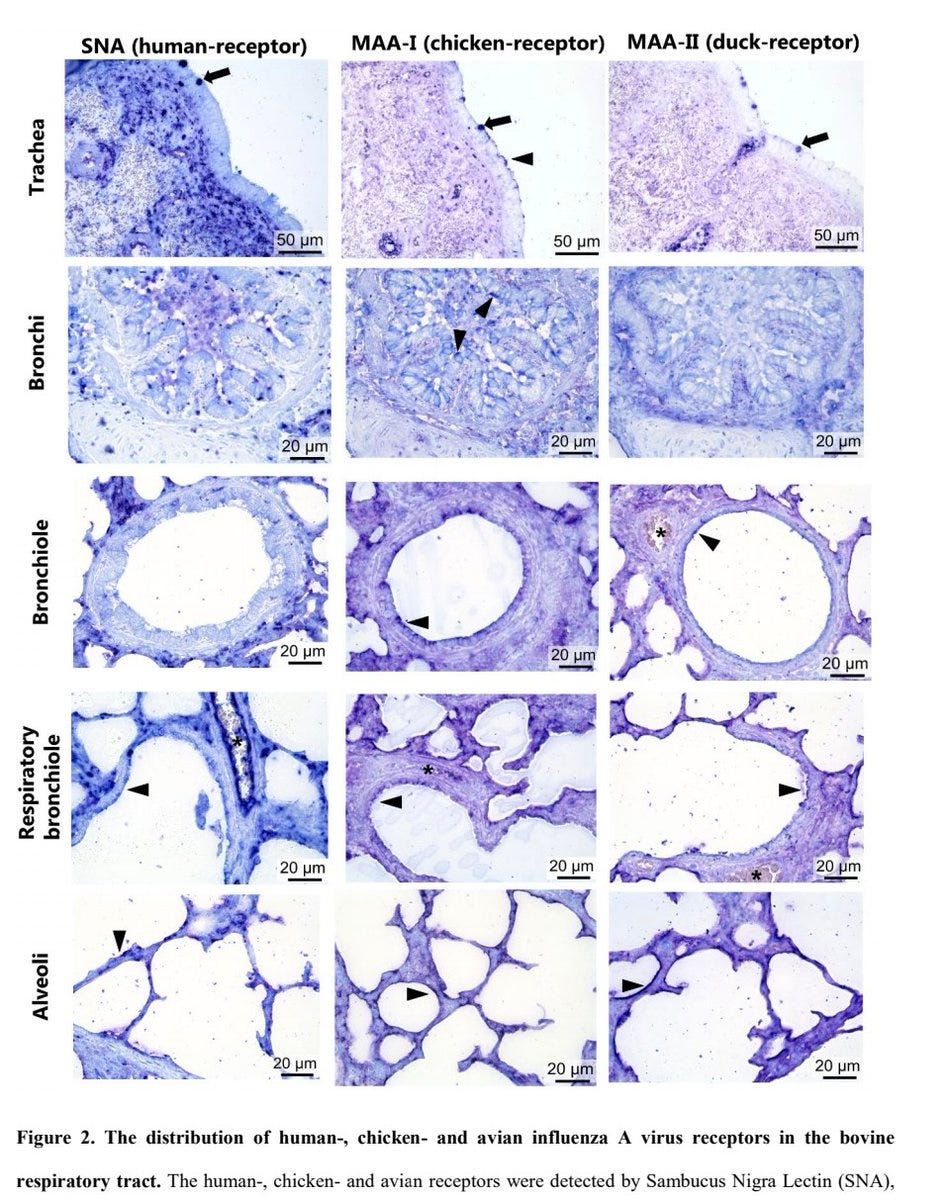
5) In respiratory tract, all receptors were expressed in trachea goblet cells. The chicken receptor was on ciliated epithelial cells. Expression decreased in lower tract.
In alveoli, all receptors were expressed. The human receptor was on type II pneumocytes/leukocytes
6) Sparse receptor expression was detected in cerebrum neurons.
Receptor expression provides rationale for high viral loads in infected bovine milk and suggests cattle could act as a mixing vessel for novel IAV generation with increased zoonotic potential.
7) In summary, this study investigated IAV receptor distribution in cattle tissues and found receptor expression patterns that help explain the recent IAV infections detected in dairy cows.
And now for something completely different …
Insect’s lives in general hang in the balance on a rather confined thermal tightrope. Desert ants (dating myself, read this when it came out) were the first to pop into my brain when I saw this. Imagine trying to live in a bee’s nest with no air conditioning. Many do build in shaded areas like trees. Turns out it is getting to be a big deal.
In a new article published in Frontiers in Bee Science, scientists identify rising heat as a potential culprit for the decline in bumblebee populations worldwide, compromising bumblebees' ability to construct livable nests in which healthy larvae can develop.
"The constraints on the survival of the bumblebee brood indicate that heat is likely a major factor, with heating of the nest above about 35 degrees Celsius being lethal, despite the remarkable capacity of bumblebees to thermoregulate."
There are many bumblebee species around the world, living in many different environments. Lots of these species are in a decline linked to climate change, but identifying a causative factor has proven difficult.
However, by reviewing the literature, Kevan and colleagues identified a critical commonality between these species, regardless of geographic range: the optimal temperature of their nests, 28–32 degrees Celsius.
Stay safe and help those who need it most.
take care,
Joe
*A special note: this is a free newsletter. I spend 4 hours a day just reading and another couple of hours putting together this newsletter. Donations out of kindness are deeply appreciated. All donations go toward the purchase of another server that will allow my modeling work to go global with a small percentage toward climate causes. You can go to my substack homepage if you wish to donate.
New addition. I love this article, easy read but stimulates the brian in many ways.
Viruses Finally Reveal Their Complex Social Life - Quanta Magazine
Where do viruses hide in the human body? | The BMJ
Understanding immunity and viruses through the John Snow Project
Variant tracking at the CDC
Infections at WHN (updated!)
The neuroinvasiveness, neurotropism, and neurovirulence of SARS-CoV-2: Trends in Neurosciences (cell.com)